Research Summary
Members
-
 Hongri Gong
Hongri Gong
professor gonghr@nankai.edu.cn
-
 Zhenwu Yu
Zhenwu Yu
postdoctor
-
 Yuying Zhang
Yuying Zhang
doctoral student
-
 Yuezheng Lai
Yuezheng Lai
doctoral student
-
 Long Yu
Long Yu
doctoral student
-
 Jinxu Xu
Jinxu Xu
doctoral student
-
 Ziyan Feng
Ziyan Feng
doctoral student
-
 Ziqing Zhao
Ziqing Zhao
doctoral student
-
 Fusen Yang
Fusen Yang
doctoral student
-
 Yue Chen
Yue Chen
doctoral student
-
 Jiliang Deng
Jiliang Deng
doctoral student -
 Yutong Liu
Yutong Liu
doctoral student
-
 Jianyun Wang
Jianyun Wang
master student
-
 Kun Shi
Kun Shi
master student
-
 Yuanyuan Zhou
Yuanyuan Zhou
master student
-
 Xiaoting Zhou
Xiaoting Zhou
Ph.D. graduate
-
 Shan Zhou
Shan Zhou
Ph.D. graduate
-
 Weiwei Wang
Weiwei Wang
Ph.D. graduate
-
 Zhanqiang Du
Zhanqiang Du
Ph.D. graduate
-
 Yanting Tang
Yanting Tang
Ph.D. graduate
Publications
-
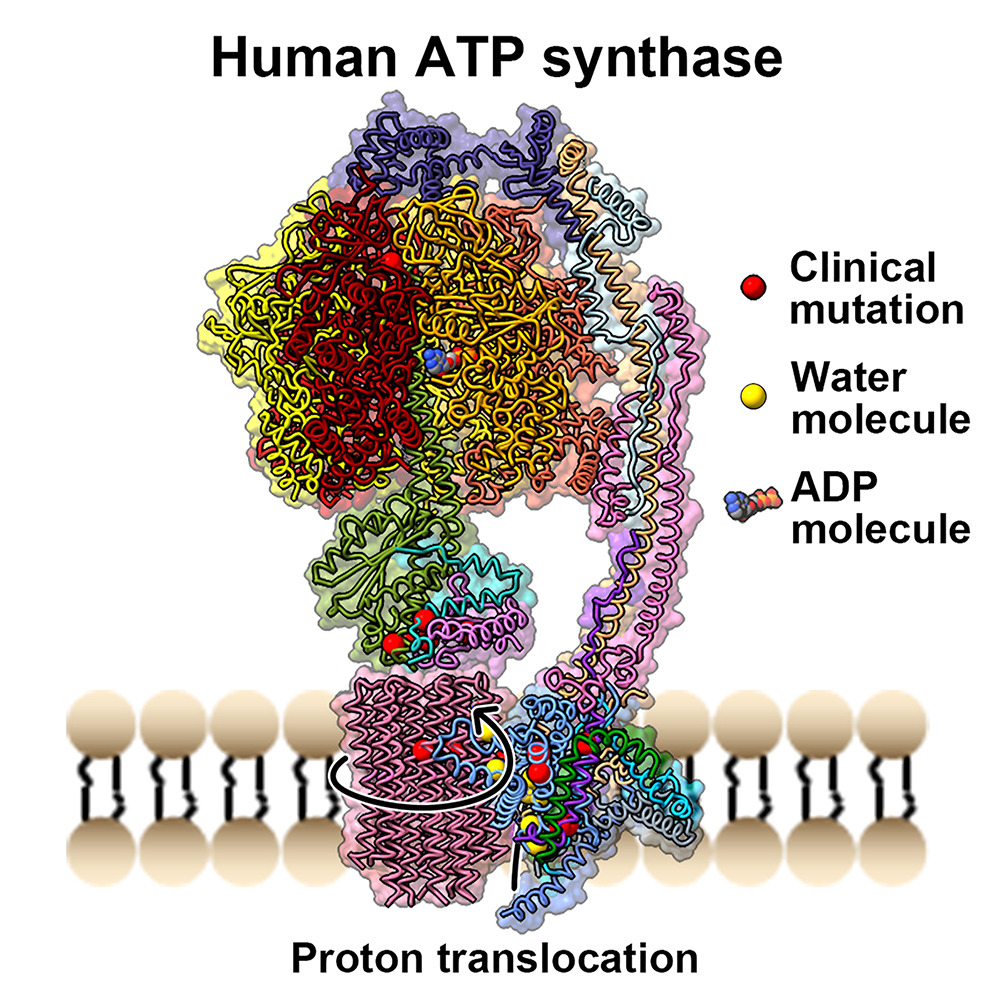
 Zhang Y#, Lai Y#, Zhou S, Ran T, Zhang Y, Zhao Z, Feng Z, Yu L, Xu J, Shi K, Wang J, Pang Y, Li L, Chen H, Guddat LW, Gao Y*, Liu F*, Rao Z*, Gong H*
Inhibition of M. tuberculosis and human ATP synthase by BDQ and TBAJ-587
Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation ofMycobacterium tuberculosisby inhibiting ATP synthase1,2. However, BDQ also inhibits human ATP synthase3. At present, how these compounds interact with eitherM. tuberculosisATP synthase or human ATP synthase is unclear. Here we pr...Nature | 2024-07-03
Zhang Y#, Lai Y#, Zhou S, Ran T, Zhang Y, Zhao Z, Feng Z, Yu L, Xu J, Shi K, Wang J, Pang Y, Li L, Chen H, Guddat LW, Gao Y*, Liu F*, Rao Z*, Gong H*
Inhibition of M. tuberculosis and human ATP synthase by BDQ and TBAJ-587
Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation ofMycobacterium tuberculosisby inhibiting ATP synthase1,2. However, BDQ also inhibits human ATP synthase3. At present, how these compounds interact with eitherM. tuberculosisATP synthase or human ATP synthase is unclear. Here we pr...Nature | 2024-07-03 -
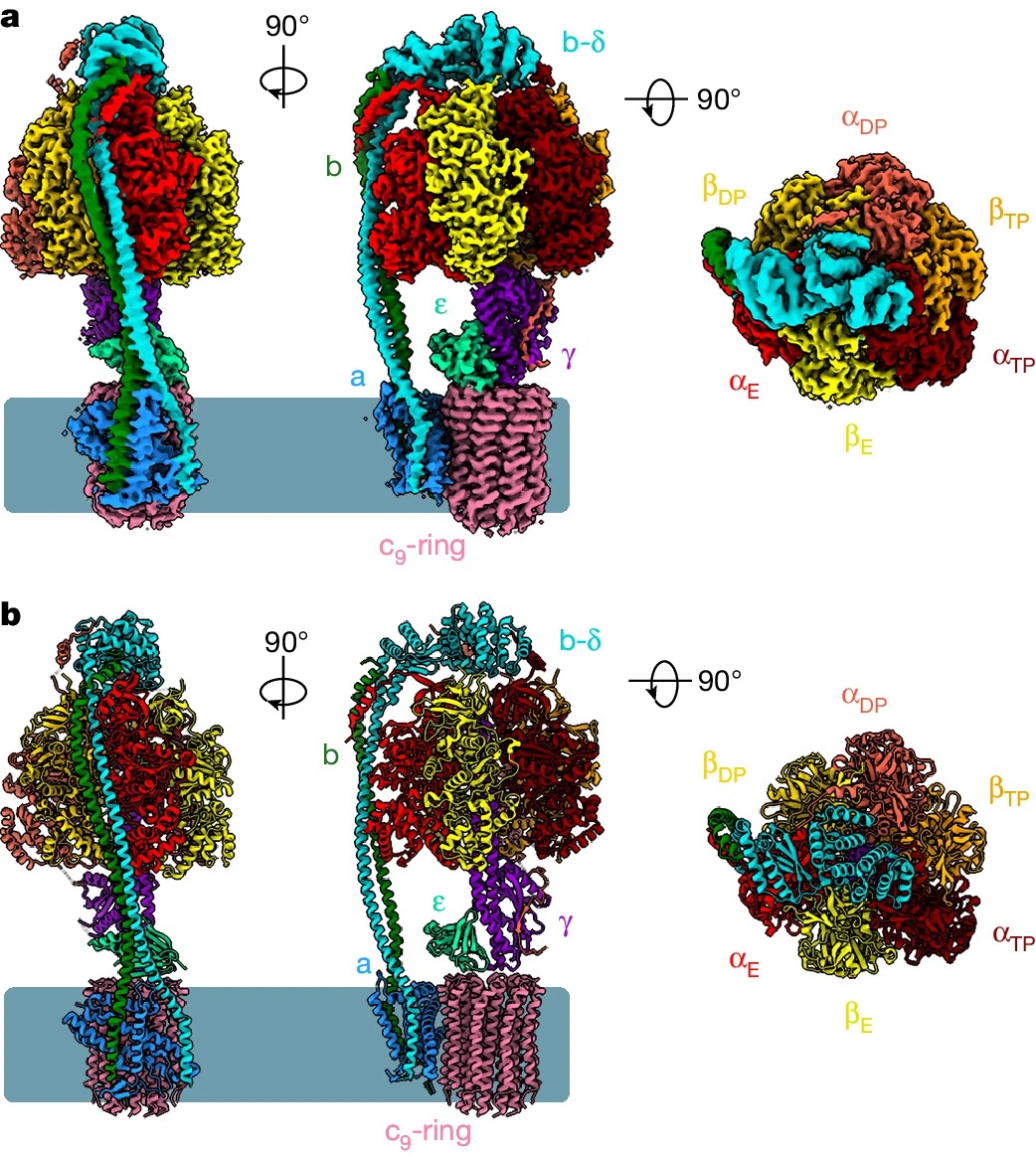
 Lai Y#, Zhang Y#, Zhou S#, Xu J#, Du Z#, Feng Z, Yu L, Zhao Z, Wang W, Tang Y, Yang X, Guddat LW, Liu F*, Gao Y*, Rao Z*, Gong H*
Structure of the human ATP synthase
Biological energy currency ATP is produced by F1Fo-ATP synthase. However, the molecular mechanism for human ATP synthase action remains unknown. Here, we present snapshot images for three main rotational states and one substate of human ATP synthase using cryoelectron microscopy. These structures reveal that the release of ADP occurs when the β subunit of F1Fo-ATP syntha...Mol Cell | 2023-05-26
Lai Y#, Zhang Y#, Zhou S#, Xu J#, Du Z#, Feng Z, Yu L, Zhao Z, Wang W, Tang Y, Yang X, Guddat LW, Liu F*, Gao Y*, Rao Z*, Gong H*
Structure of the human ATP synthase
Biological energy currency ATP is produced by F1Fo-ATP synthase. However, the molecular mechanism for human ATP synthase action remains unknown. Here, we present snapshot images for three main rotational states and one substate of human ATP synthase using cryoelectron microscopy. These structures reveal that the release of ADP occurs when the β subunit of F1Fo-ATP syntha...Mol Cell | 2023-05-26 -
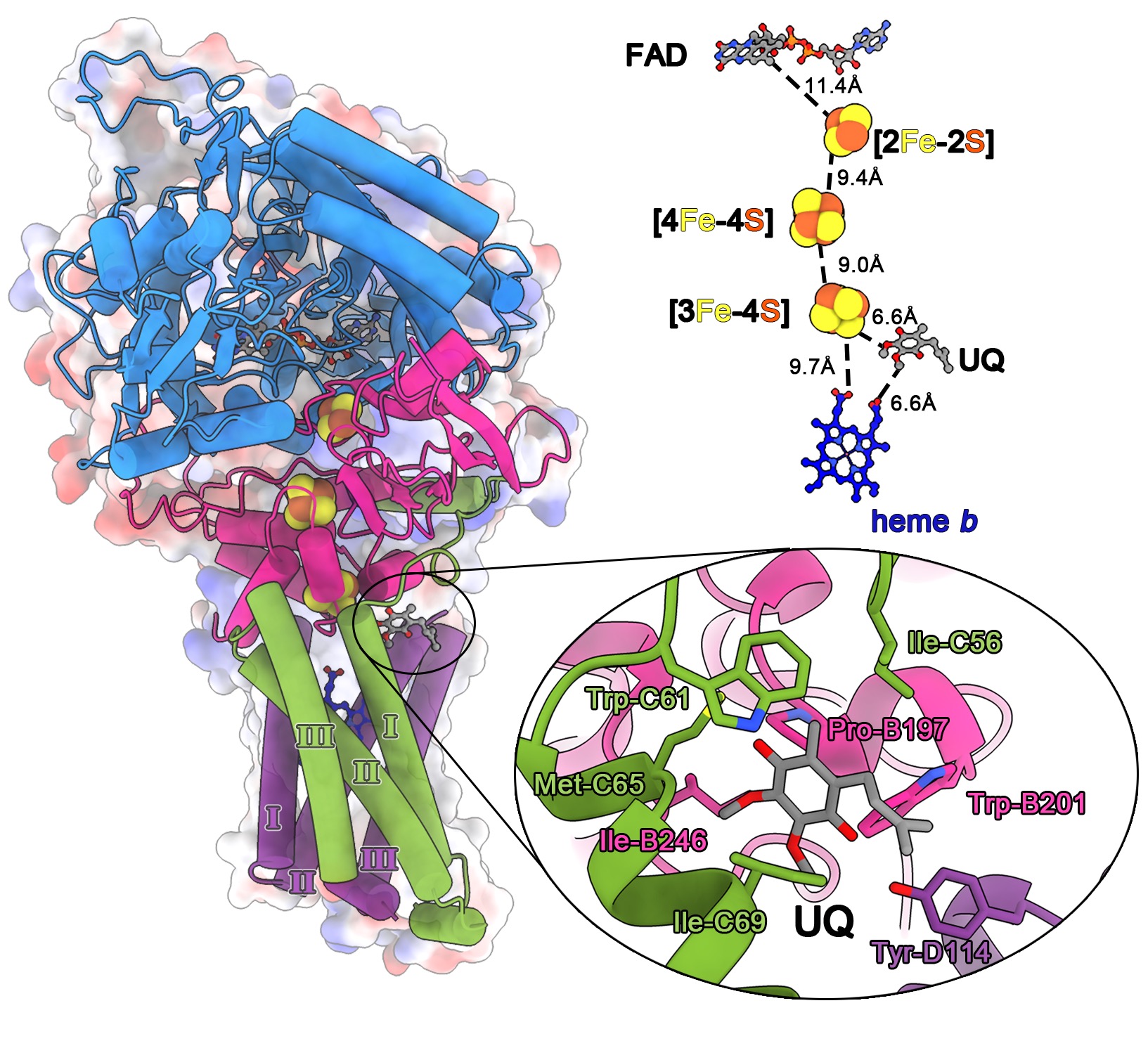 Du Z#, Zhou X#, Lai Y, Xu J, Zhang Y, Zhou S, Feng Z, Yu L, Tang Y, Wang W, Yu L, Tian C, Ran T, Chen H, Guddat LW, Liu F*, Gao Y*, Rao Z*, Gong H*
Structure of the human respiratory complex II
Human complex II is a key protein complex that links two essential energy-producing processes: the tricarboxylic acid cycle and oxidative phosphorylation. Deficiencies due to mutagenesis have been shown to cause mitochondrial disease and some types of cancers. However, the structure of this complex is yet to be resolved, hindering a comprehensive understanding of the functional aspects of thi...Proc Natl Acad Sci U S A | 2023-04-25
Du Z#, Zhou X#, Lai Y, Xu J, Zhang Y, Zhou S, Feng Z, Yu L, Tang Y, Wang W, Yu L, Tian C, Ran T, Chen H, Guddat LW, Liu F*, Gao Y*, Rao Z*, Gong H*
Structure of the human respiratory complex II
Human complex II is a key protein complex that links two essential energy-producing processes: the tricarboxylic acid cycle and oxidative phosphorylation. Deficiencies due to mutagenesis have been shown to cause mitochondrial disease and some types of cancers. However, the structure of this complex is yet to be resolved, hindering a comprehensive understanding of the functional aspects of thi...Proc Natl Acad Sci U S A | 2023-04-25 -
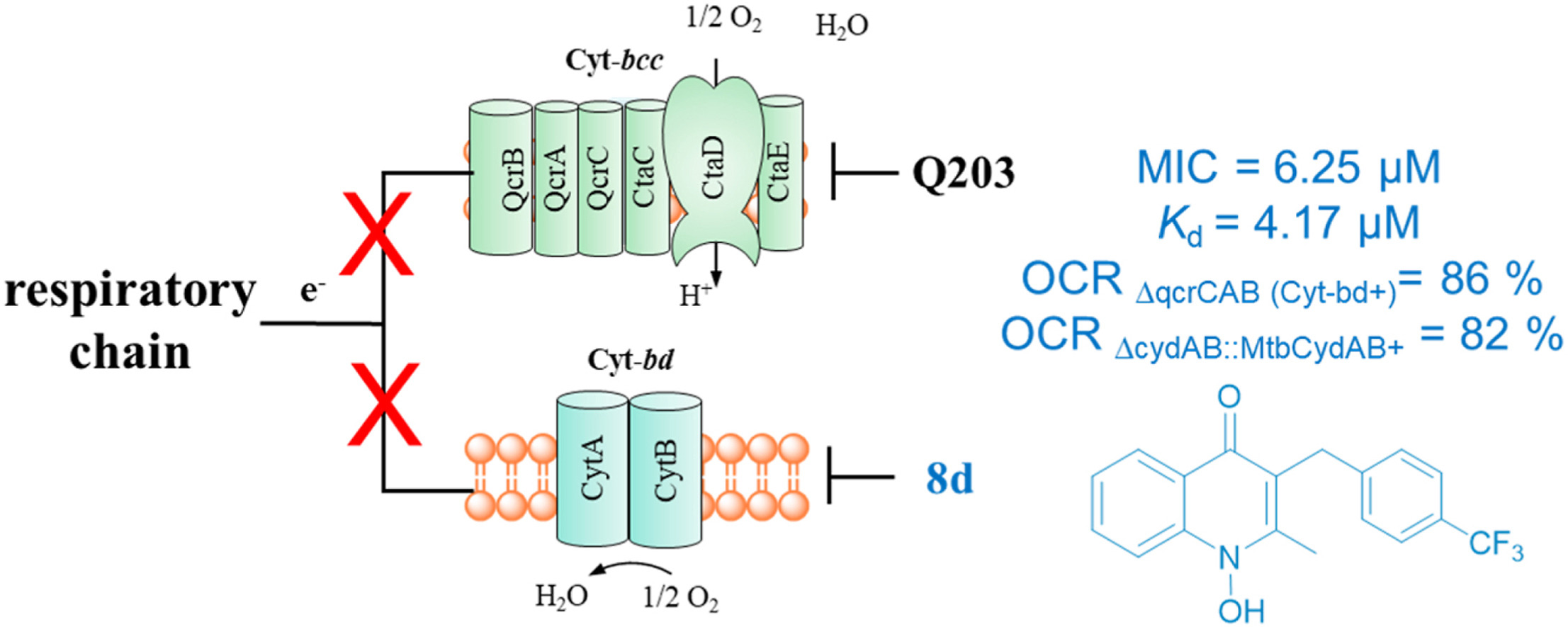 Zhou Y#, Shao M#, Wang W#, Cheung CY, Wu Y, Yu H, Hu X, Cook GM, Gong H*, Lu X*
Discovery of 1-hydroxy-2-methylquinolin-4(1H)-one derivatives as new cytochrome bd oxidase inhibitors for tuberculosis therapy
The cytochrome bcc-aa oxidase (Cyt-bcc) of Mycobacterium tuberculosis (Mtb) is a promising anti-tuberculosis target. However, when3 Cyt-bcc is inhibited, cytochrome bd terminal oxidase (Cyt-bd) can still maintain the activity of the respiratory chain and drive ATP synthesis. Through virtual screening and biological validation, we discovered two FDA-approved drugs, ivacaftor and roqui...Eur J Med Chem | 2023-01-05
Zhou Y#, Shao M#, Wang W#, Cheung CY, Wu Y, Yu H, Hu X, Cook GM, Gong H*, Lu X*
Discovery of 1-hydroxy-2-methylquinolin-4(1H)-one derivatives as new cytochrome bd oxidase inhibitors for tuberculosis therapy
The cytochrome bcc-aa oxidase (Cyt-bcc) of Mycobacterium tuberculosis (Mtb) is a promising anti-tuberculosis target. However, when3 Cyt-bcc is inhibited, cytochrome bd terminal oxidase (Cyt-bd) can still maintain the activity of the respiratory chain and drive ATP synthesis. Through virtual screening and biological validation, we discovered two FDA-approved drugs, ivacaftor and roqui...Eur J Med Chem | 2023-01-05 -
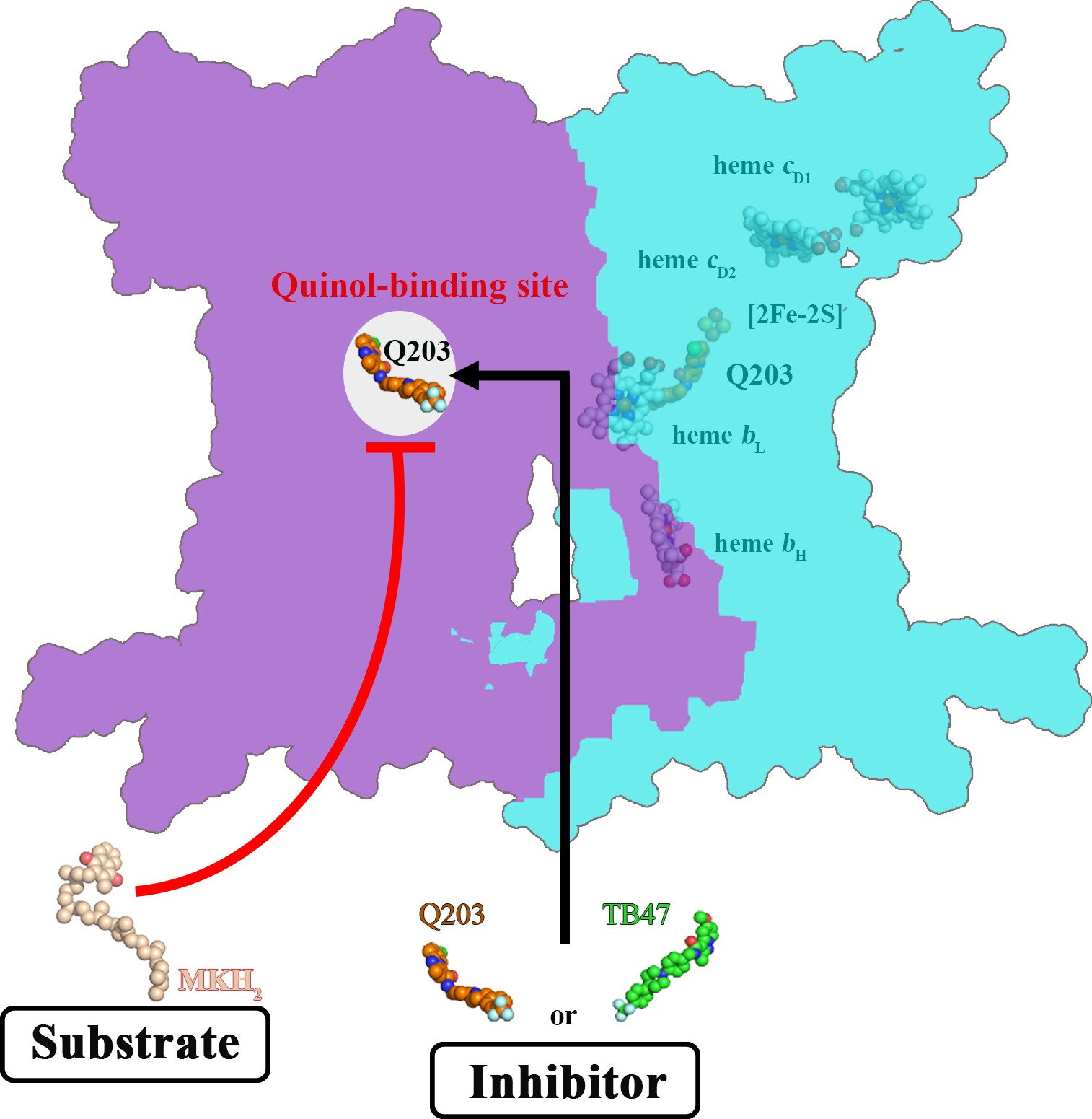 Zhou S#, Wang W#, Zhou X, Zhang Y, Lai Y, Tang Y, Xu J, Li D, Lin J, Yang X, Ran T, Chen H, Guddat LW, Wang Q, Gao Y*, Rao Z* & Gong H*
Structure of Mycobacterium tuberculosis cytochrome bcc in complex with Q203 and TB47, two anti-TB drug candidates
Pathogenic mycobacteria pose a sustained threat to global human health. Recently, cytochrome bcc complexes have gained interest as targets for antibiotic drug development.These high-resolution images provide a basis for the design of new mycobacterial cytElife | 2021-11-25
Zhou S#, Wang W#, Zhou X, Zhang Y, Lai Y, Tang Y, Xu J, Li D, Lin J, Yang X, Ran T, Chen H, Guddat LW, Wang Q, Gao Y*, Rao Z* & Gong H*
Structure of Mycobacterium tuberculosis cytochrome bcc in complex with Q203 and TB47, two anti-TB drug candidates
Pathogenic mycobacteria pose a sustained threat to global human health. Recently, cytochrome bcc complexes have gained interest as targets for antibiotic drug development.These high-resolution images provide a basis for the design of new mycobacterial cytElife | 2021-11-25 -
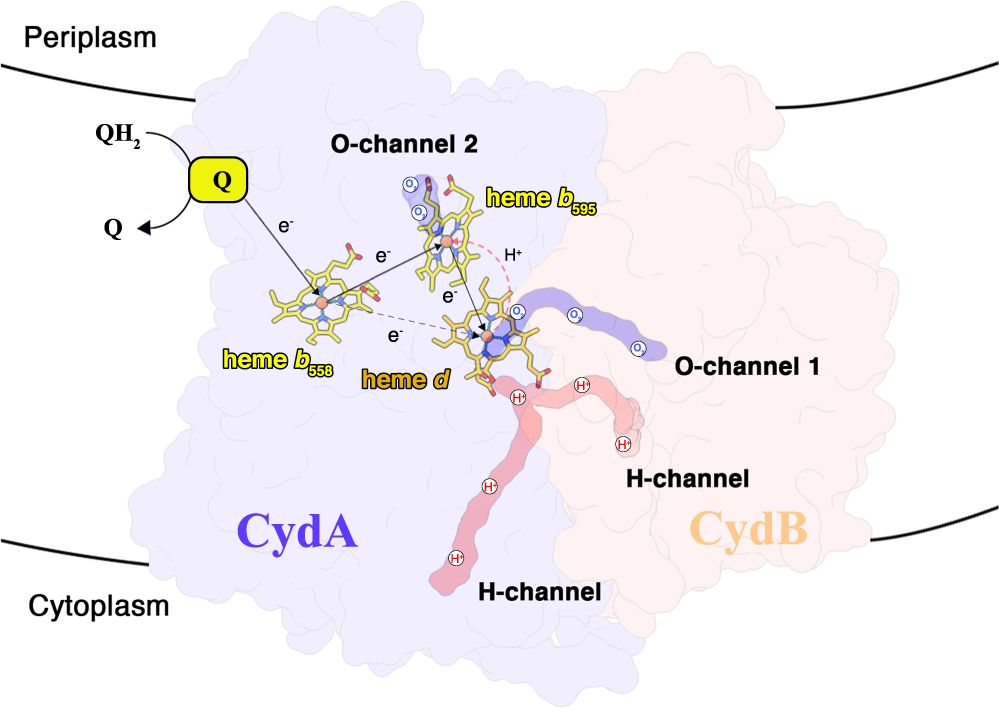 Wang W#, Gao Y#, Tang Y, Zhou X, Lai Y, Zhou S, Zhang Y, Yang X, Liu F, Guddat LW, Wang Q*, Rao Z* & Gong H*
Cryo-EM structure of mycobacterial cytochrome bd reveals two oxygen access channels
Cytochromes bd are ubiquitous amongst prokaryotes including many human-pathogenic bacteria. Such complexes are targets for the development of antimicrobial drugs. However, an understanding of the relationship between the structure and functional mechanismNat Commun | 2021-07-30
Wang W#, Gao Y#, Tang Y, Zhou X, Lai Y, Zhou S, Zhang Y, Yang X, Liu F, Guddat LW, Wang Q*, Rao Z* & Gong H*
Cryo-EM structure of mycobacterial cytochrome bd reveals two oxygen access channels
Cytochromes bd are ubiquitous amongst prokaryotes including many human-pathogenic bacteria. Such complexes are targets for the development of antimicrobial drugs. However, an understanding of the relationship between the structure and functional mechanismNat Commun | 2021-07-30